|
| | lenvatinib Mesylate Basic information |
| Product Name: | lenvatinib Mesylate | | Synonyms: | lenvatinib Methanesulfonate;E7080 Mesylate;4-[3-Chloro-4-[[(cyclopropylamino)carbonyl]amino]phenoxy]-7-methoxy-6-quinolinecarboxamide monomethanesulfonate;4-[3chloro-4-(N'-cyclopropylureido)phenoxy]-7-methoxyquinoline-6-carboxamide methanesulfonate;CAT#A863437;Lenvatinib Mesylate,Amadis Chemical offer CAS#857890-39-2;lenvatinib Mesylate;E7080;E-7080;E 7080 | | CAS: | 857890-39-2 | | MF: | C22H23ClN4O7S | | MW: | 522.95862 | | EINECS: | 812-398-0 | | Product Categories: | FGF receptor antagonist;API;API;857890-39-2;chemical | | Mol File: | 857890-39-2.mol |  |
| | lenvatinib Mesylate Chemical Properties |
| Melting point | >220°C (dec.) | | storage temp. | -20°C Freezer | | solubility | DMSO (Slightly), Methanol (Slightly) | | form | Solid | | color | White to Off-White |
| | lenvatinib Mesylate Usage And Synthesis |
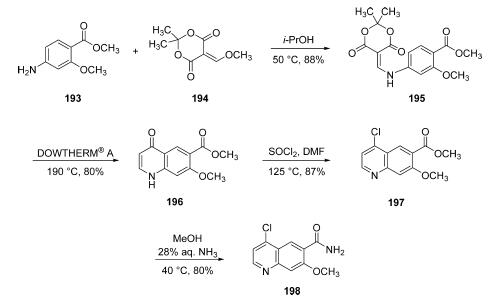
| Description | Lenvatinib mesylate (lenvatinib) is an orally available, receptor‐type tyrosine kinase inhibitor, which was developed at Eisai in 2015. It was approved by the FDA in 2015 for the treatment of differentiated thyroid cancer that is either locally recurrent, metastatic, or progressive and did not respond to radioactive iodine treatment. In May 2016, the FDA approved the drug as a combination therapy with everolimus for the treatment of advanced renal cell carcinoma. Because VEGF (and fibroblast growth factor receptors, known as FGFRs) are thought to play a role in cardiovascular signaling pathways, VEGF2R and FGFR inhibition are thought to be the mechanisms behind the primary side effect of lenvatinib mesylate, which is hypertension. | | Uses | E7080 (Lenvatinib) is a multi-target inhibitor of VEGFR2 and VEGFR3 with IC50 of 4 nM and 5.2 nM, respectively. | | Uses | Lenvatinib Mesylate is used in preparation of anti-human CTLA4xPD-1 bispecific antibodies for diagnosis, prevention and treatment of tumor or anemia. | | Definition | ChEBI: Lenvatinib mesylate is a methanesulfonate salt obtained by reaction of lenvatinib with one molar equivalent of methanesulfonic acid. A multi-kinase inhibitor and orphan drug used (as its mesylate salt) for the treatment of various types of thyroid cancer that do not respond to radioiodine. It has a role as an EC 2.7.10.1 (receptor protein-tyrosine kinase) inhibitor, a fibroblast growth factor receptor antagonist, an orphan drug, a vascular endothelial growth factor receptor antagonist and an antineoplastic agent. It contains a lenvatinib(1+). | | Synthesis | Starting from commercial aniline
193, a substitution reaction under neutral conditions in warm
isopropyl alcohol with a commercial vinyl methoxy derivative of
Meldrum?ˉs acid (194) produced enamine 195 in good yield.
Next, subjection of 195 to DOWTHERM A at 190 ??C affected
an intramolecular cyclizative substitution reaction, followed by
loss of acetone, and a decarboxylation reaction to furnish
quinolone 196. This cyclization reaction, which is a variant of
the Conrad-Limpach reaction, is particularly noteworthy
given the temperature and pH at which it takes place. Conrad-
Limpach cyclizations typically proceed under basic conditions
at temperatures well above 240 ??C. However, a process was
developed by Zeneca in 2004 which involved subjecting 195 to
the DOWTHERM heat transfer fluid (commercially available
from Dow and Sigma-Aldrich, consisting of a eutectic mixture
of biphenyl and diphenyl oxide) allowed the team to lower
the temperature required for the reaction, clearly observe
bubbling of gas indicating the progress of the reaction, and
simple cooling and treatment with ether to facilitated
precipitate formation. The resulting solid could be collected
by filtration and required no additional purification on scale in
80% yield. Quinoline 196 was then converted to the
corresponding chloride using thionyl chloride in refluxing
DMF, and the resulting ester 197 was converted to the
corresponding amide through the use of 28% aqueous
ammonia in warm ethanol, which ultimately produced the
key chloroquinoline lenvatinib subunit 198 in 80% yield from
197.
Commercial aminophenol 199 was
converted to the corresponding carbamate through the use of
phenyl chloroformate in essentially quantitative yield prior to
subjection to cyclopropylamine in chilled DMF, which
ultimately furnished urea 201 in 77% overall yield from 200.
Next, exposure of phenol 201 to chloroquinoline 198 in the presence of potassium t-butoxide followed by
treatment with methanesulfonic acid and acetic acid resulted in
clean formation of lenvatinib mesylate (XXV) in 96% yield
across the two-step sequence.
 |
| | lenvatinib Mesylate Preparation Products And Raw materials |
|




