|
| | 2,4-Dichlorophenoxyacetic acid Chemical Properties |
| Melting point | 136-140 °C (lit.) | | Boiling point | 160 °C (0.4 mmHg) | | density | 1.563 | | vapor pressure | 0.4 mm Hg ( 160 °C) | | refractive index | 1.5000 (estimate) | | Fp | 160°C/0.4mm | | storage temp. | 2-8°C | | solubility | Soluble in organic solvents (ethanol, acetone, dioxane) | | form | crystalline | | pka | pK1:2.64 (25°C) | | color | off-white to tan | | PH Range | Acidic | | Odor Threshold | 3.13 ppm | | Water Solubility | Slightly soluble. Decomposes. 0.0890 g/100 mL | | Merck | 14,2796 | | BRN | 1214242 | | Henry's Law Constant | 6.72 and 0.84 x 10-5 atm?m3/mol were reported at pH values of 1 and 7, respectively (wetted-wall column, Rice et al.,
1997a) | | Exposure limits | NIOSH REL: TWA 10 mg/m3, IDLH 100 mg/m3; OSHA PEL: TWA
10 mg/m3; ACGIH TLV: TWA 10 mg/m3.
| | Stability: | Stable, but moisture-sensitive and may be light-sensitive. Incompatible with strong oxidizing agents, corrodes many metals. Decomposes in water. | | LogP | 2.810 | | CAS DataBase Reference | 94-75-7(CAS DataBase Reference) | | IARC | 2B (Vol. 113) 2018 | | NIST Chemistry Reference | (2,4-Dichlorophenoxy)acetic acid(94-75-7) | | EPA Substance Registry System | 2,4-D (94-75-7) |
| | 2,4-Dichlorophenoxyacetic acid Usage And Synthesis |
| Description | As one of the most inexpensive and oldest weed killers, 2,4-Dichlorophenoxyacetic acid (usually called 2,4-D) is a systemic herbicide that is widely applied all over the world. It is effective to selectively kill a variety of terrestrial and aquatic broadleaf weeds without affecting most grasses, such as cereals, lawn turf, and grassland. Nowadays, 2,4-D is widely used to treat unwanted vegetation in various areas. In the field of forestry, it is used for stump treatment, trunk injection, selective control of brush in conifer forests and used to kill weeds and brush along roadways, railways, and power lines which probably damage equipment or interfere with safe operation. Besides, it is applied to control aquatic weeds for the safety of boating, fishing and swimming or protection of hydroelectric equipment. It is also used to control the spread of invasive, noxious and non-native weed species by government and control various poisonous weeds such as poison ivy and poison oak. In addiction to the forestry use, 2,4-D can also be used as a supplement in plant cell culture media in laboratories as a dedifferentiation hormone.
| | References | https://www.nrdc.org/stories/24-d-most-dangerous-pesticide-youve-never-heard
https://pubchem.ncbi.nlm.nih.gov/compound/1486#section=Top
https://en.wikipedia.org/wiki/2,4-Dichlorophenoxyacetic_acid
| | Description | 2,4-D free acids, esters, amines, and salts are formulated in
water suspensions or solutions, or in various organic solvents,
for application as systemic herbicides that are used postemergence
for selective control of broadleaf weeds. | | Chemical Properties | 2,4-Dichlorophenoxyacetic acid is a white to yellow crystalline powder with a slight phenolic odor. It is used as a herbicide for the selective control of broad-leaved weeds in agriculture, and for the control of woody plants along roadsides, railways, and utilities rights of way. It is one of the most widely used herbicides in the world and is commonly used on crops such as wheat and corn, and on pasture and rangelands. It is also used to control broad-leaved aquatic weeds.
| | Physical properties | Odorless, white to pale yellow, powder or prismatic crystals. Impure formulations containing 2,4-
D as the main component may have a phenolic odor. | | Uses | 2,4-D is
registered with the US Environmental Protection Agency (EPA)
for use on a variety of food/feed sites, turf, lawn, aquatic sites,
and forestry applications, and as a growth regulator in citrus
crops. Residents and professional applicators may use 2,4-D on
home lawns. | | Uses | 2,4-Dichlorophenoxyacetic acid is often formulated as various
forms of inorganic salts or esters. 2,4-D was first registered
as a herbicide in 1948, and its annual production was
estimated at 52–67 million lb in 1990. The primary use
of 2,4-D is for control of broadleaf weeds, and as such, it is
used for a large spectrum of applications in agriculture,
forestry, and lawn care. 2,4-D also is used along right-ofways,
on rangelands, parks, and in aquatic environments. | | Uses | 2,4-D is one of the most widely used herbicides. It is a chlorine-substituted phenoxyacetic acid herbicide used for postemergence control of annual and perennial broad-leaved weeds in fruits, vegetables, turfs and ornamentals.Exposure to this chemical may arisein the garden, farm, or plantation setting,where it is sprayed for weed control andto enhance the plant growth. Its residueshave been detected in soils, sediments, andgroundwater, as well as in tissues of corals(Crockett et al. 1986; Glynn et al. 1984). | | Preparation | Two processes are currently used for the production of 2,4-Dichlorophenoxyacetic acid. In the first process, phenol is condensed with chloroacetic acid forming phenoxyacetic acid, which is subsequently chlorinated. In the second process, phenol is chlorinated, generating 2,4-dichlorophenol, which is subsequently condensed with chloroacetic acid. The butyl ester derivative of 2,4-D is produced by the esterification of the acid with butanol in the presence of a ferric chloride catalyst and chlorine (Liu et al., 2013).
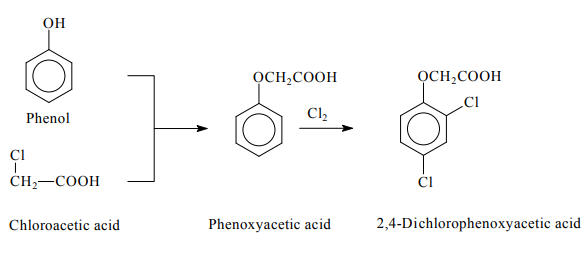
Preparation of 2,4-Dichlorophenoxyacetic acid
| | Definition | ChEBI: A chlorophenoxyacetic acid that is phenoxyacetic acid in which the ring hydrogens at postions 2 and 4 are substituted by chlorines. | | General Description | Odorless white to tan solid. Sinks in water. | | Air & Water Reactions | Decomposes rapidly in water. | | Reactivity Profile | 2,4-Dichlorophenoxyacetic acid is incompatible with strong oxidizers. 2,4-Dichlorophenoxyacetic acid is corrosive to metals. | | Health Hazard | The acute toxic symptoms from ingestion,absorption through skin, and inhalation ofits dusts include lethargy, stupor, weakness,incoordination, muscular twitch, nausea, vomiting, gastritis, convulsions, and coma.Death resulted in animals from ventricularfibrillation. In humans, ingestion of 4–6 gof 2,4-D can cause death. The toxic effectsare nausea, vomiting, somnolence, convul-sions, and coma. The oral LD50 value in testanimals varied at 100–500 mg/kg, depend-ing on species, which included dogs, guineapigs, hamsters, rats, mice, and rabbits.
Gorzinski and co-workers (1987) have investigated acute, pharmacokinetic, and subchronic toxicity of 2,4-D and its estersand salts in rats and rabbits. The acutedermal LD50 values in rabbits for theacid, esters, and salts were >2000 mg/kg.Urinary elimination of 2,4-D was saturatedin male rats given oral doses of 50 mg/kg.There was a decrease in body weightgain and increase in kidney weightsin animals, resulting from subchronicdietary doses. A dose-related degenerativechange was observed in the proximalrenal tubules. Repeated subcutaneous dosing(150–250 mg/kg) of 2,4-D butyl esterresulted in accumulation of the compoundin the brain, causing neurobehavioral toxicity(Schulze and Dougherty 1988).
Alexander and associates (1985) determined the 48-hour median lethal con-centration values for 2,4-D to aquatic organ-isms. These values are 25, 325, 290, and358 mg/L for Daphnia magna, fathead min-nows, bluegill, and rainbow trout, respec-tively.
| | Fire Hazard | Special Hazards of Combustion Products: Toxic and irritating hydrogen chloride or phosgene gases may form. | | Agricultural Uses | Herbicide, Plant growth regulator: 2,4-Dichlorophenoxyacetic acid was introduced as a plant growth-regulator in 1942. 2, 4-D is the most widely used herbicide in the United States and its used in more than 100 countries. It is registered in the United States as a herbicide for control of broadleaf plants and as a plant growth-regulator. There are many forms or derivatives of 2,4-D including esters, amines, and salts. It is used in cultivated agriculture, in pasture and rangeland applications, forest management, home, garden, and to control aquatic vegetation. It may be found in emulsion form, in aqueous solutions (salts), and as a dry compound. The product Agent Orange, made by Monsanto Chemical and used extensively throughout Vietnam, was about 50% 2,4-D. However, the controversies associated with the use of Agent Orange involved a contaminant (dioxin) in the 2,4,5-T component of the defoliant. In 1964 Agent Orange replaced Agent Purple a mixture of the n-butyl esters of 2,4-D and 2,4,5-T plus the isobutyl ester of 2,4,5-T. | | Trade name | Hedonal; 2,4-D; Estone; Agrotect; Fernesta; Fernimine; Netagrone; Tributon; Vergemaster; Amoxone; Dicopur; Dormone; Ipaner; Moxone; Phenox; Pielik; Rhodia; Weedone; B-Selektonon. | | Potential Exposure | 2,4-Dichlorophenoxyacetic acid,
was introduced as a plant growth-regulator in 1942. It
is registered in the United States as a herbicide for control
of broadleaf plants and as a plant growth-regulator. Thus,
workers engaged in manufacture, formulation or application
are affected, as may be citizens in areas of application. The
Vietnam war era defoliant, Agent Orange, was a mixture of
2,4-D and 2,4,5-T. | | Carcinogenicity | Collectively, the epidemiological
and toxicological data show that 2,4-D is not likely to be
carcinogenic in humans unless it is acting through an
unknown mechanism that is not evident in animals. According
to the calculated RfD and data from exposure studies, the
general public should not experience toxic effects from
exposure to 2,4-D. Because workers involved in the manufacture
or application of 2,4-D may be exposed to levels
above the RfD, appropriate protective equipment should be
used. | | Environmental Fate | 2,4-Dichlorophenoxyacetic acid (2,4-d) is rapidly degraded by microbes in soil and water, with a half-life of 3-22 days in different soils. 2,4-d is weakly sorbed by soil with sorption generally increasing with increasing soil organic carbon content. Leaching to groundwater is most likely in coarse-grained sandy soils with low organic content or with very basic soils. In general, little runoff occurs with 2,4-d or its amine salts. | | Metabolism | Chemical. 2,4-D and its salts are very stable, but
esters are sensitive to hydrolysis under acidic and basic conditions. In the field, 2,4-D losses due to
photodegradation are minor. 2,4-D is a strong acid and
forms water-soluble salts with amines and alkali metals.
A sequestering agent is included in 2,4-D formulations to
prevent precipitation of Ca2+ andMg2+ salts in hard water.
Plant. 2,4-D detoxification occurs relatively slowly in
plants. There are many possible routes of detoxification,
and these are usually grouped into those reactions that are
consistent with phase I metabolism and those that are consistent
with phase II metabolism. Phase I reactions that
have been observed to occur with 2,4-D include dechlorination,
decarboxylation, hydroxylation, and dealkylation.
Phase II reactions that have been observed to occur
with 2,4-D include conjugation of the side chain to
amino acids, particularly glutamate and aspartate, and glucose conjugation following hydroxylation of the phenoxy
ring. Selectivity differences among broadleaf species
may be accounted for by differences in the rates of 2,4-D
detoxification.
Soil. Microbial degradation in the soil involves cleavage
of the acid side chain, decarboxylation, hydroxylation, and
ring opening. | | Shipping | UN3345 Phenoxyacetic acid derivative pesticide,
solid, toxic, Hazard Class: 6.1; Labels: 6.1-Poisonous
materials. A DOT regulated marine pollutant. | | Purification Methods | Crystallise 2,4-D from MeOH. It is a plant growth substance, a herbicide and is TOXIC. [Beilstein 6 IV 908.] | | Toxicity evaluation | 2,4-D is excreted unchanged in
the urine of humans and rat. When used under normal
conditions, 2,4-D does not appear to produce acute toxic
effects on any animal species. The acute oral LD50s of 2,4-
D in rat and mouse are 639–764 mg/kg, and 138 mg/kg,
respectively. | | Incompatibilities | A weak acid, incompatible with bases.
Decomposes in sunlight or heat, forming hydrogen chloride
and phosgene. Contact with strong oxidizers may cause fire
and explosions. | | Waste Disposal | Incineration of phenoxys is effective in 1 second @ 982 C, using a straight combustion process or @ 482℃ using catalytic combustion. Over 99% decomposition was reported when small amounts of 2,4-D were burned in a polyethylene bag. See "References" for additional detail. In accordance with 40CFR165, follow (31); recommendations for the disposal of pesticides and pesticide containers. Must be disposed properly by follow- ing (100) Package (2) label directions or by contacting your local or federal environmental control agency, or by contacting your regional EPA office. Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal.
|
| | 2,4-Dichlorophenoxyacetic acid Preparation Products And Raw materials |
|



