| | 4,4'-Methylenedianiline Chemical Properties |
| Melting point | 89-91 °C(lit.) | | Boiling point | 242 °C2 mm Hg(lit.) | | density | 1.15 | | vapor pressure | 0Pa at 25℃ | | refractive index | 1.5014 (estimate) | | Fp | 430 °F | | solubility | water: soluble | | form | neat | | pka | 5.32±0.25(Predicted) | | color | White to Light yellow | | Water Solubility | Slightly soluble. <0.1 g/100 mL at 19 ºC | | Merck | 14,2980 | | BRN | 474706 | | Exposure limits | ACGIH: TWA 0.1 ppm (Skin)
OSHA: STEL 100 ppb | | LogP | 1.55 at 25℃ | | CAS DataBase Reference | 101-77-9(CAS DataBase Reference) | | IARC | 2B (Vol. 39, Sup 7) 1987 | | NIST Chemistry Reference | Benzenamine, 4,4'-methylenebis-(101-77-9) | | EPA Substance Registry System | 4,4'-Methylenedianiline (101-77-9) |
| | 4,4'-Methylenedianiline Usage And Synthesis |
| Chemical Properties | 4,4'-Methylenedianiline is a light brown crystalline solid with a faint amine odor. lt is very slightly soluble in water and soluble in alcohol, benzene,and ether. lt is combustible when exposed to heat or flame. When heated to decomposition,it emits toxic fumes of aniline and nitrogen oxides (NOx). The dihydrochloride is a crystalline solid that is soluble in water.

4,4'-Methylenedianiline is primarily used to produce 4,4-'methylenedianiline diisocyanate and other polymeric isocyanates which are used to manufacture polyurethane foams.
| | Uses | 4,4'-Methylenedianiline is used in the production of polyurethane foams and epoxy resins. Potential contributor in pulmonary arterial hypertension (PAH) in rats through alteration of the serotenergic transport system. Drinking water contaminant candidate list 3 (CCL 3) compound as per United States Environmental Protection Agency (EPA), environmental, and food contaminants. Dyes and metabolites, Environmental Testing. | | Preparation | 93 g (1 mole) of aniline are dissolved in 96 % alcohol and to the cooled solution with proper stirring 120 g (1 mole) of 40 % formaldehyde solution are gradually added. After the addition is completed a second molecular portion of aniline, 93 g (1 mole) of aniline are added and the reaction is continued until the odor of formaldehyde has disappeared. The complete the reaction the mixture is refluxed for 2 hours. 130 g (1 mole) of aniline hydrochloride are then added, and boiling continued for a further 12 hours. The alcohol is distilled off, the residue made alkaline with the solution of sodium hydroxide and the excess of aniline removed by distillation with superheated steam. The residual oil, which consists of 4,4′-diaminodiphenylmethane, is then purified by solution in dilute hydrochloric acid and reprecipitation with dilute alkali. The 4,4′-diaminodiphenylmethane base is filtered off, washed and dried.
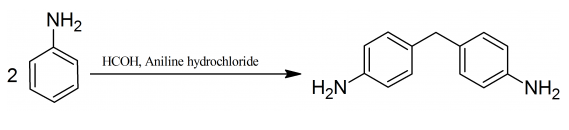
Organic medical chemicals, by M. Barrowliff, 188-189, 1921
| | Description | 4,4'-Methylenedianiline is an industrial chemical that is not known to occur naturally. It is also commonly known as diaminodiphenylmethane or MDA. It occurs as a colorless to pale yellow solid and has a faint odor. 4,4'-Methylenedianiline is used mainly for making polyurethane foams, which have a variety of uses, such as insulating materials in mailing containers. It is also used for making coating materials, glues, Spandex? fiber, dyes, and rubber. 4,4'-Methylenedianiline is also a by-product of azo dyes. It is also possibly formed by hydrolysis of diphenylmethane-4A'-diisocyanate. | | Chemical Properties | 4,4-Diaminodiphenylmethane is a pale yellow crystalline solid (turns light brown on contact with air) with a faint amine-like odor that is unstable in the presence of light or air and emits toxic fumes of aniline and nitrogen oxides when heated to decomposition. 4,4'-Methylenedianiline is primarily used in industry as a chemical intermediate in the production of 4,4-methylenedianiline diisocyanates and polyisocyanates, but is also used as a cross-linking agent for the determination of tungsten and sulfates, and as a corrosion inhibitor. Exposure to this substance irritates the skin and eyes and causes liver damage. 4,4'-Methylenedianiline is reasonably anticipated to be a human carcinogen. (NCI05) | | Uses | 4,4'-methylenedianiline be used as organic intermediates. Mainly used for the synthesis of polyimide and as curing agent of epoxy resin. | | Uses | As chemical intermediate in production of isocyanates and polyisocyantes for preparation of polyurethane foams, Spandex fibers; as curing agent for epoxy resins and urethane elastomers; in production of polyamides; in the determination of tungsten and sulfates; in preparation of azo dyes; as corrosion inhibitor. | | Uses | 4,4'-Diaminodiphenyl-methane is used in the determination of tungsten and sulfates; in the preparation of azo dyes; cross-linking agent for epoxy resins; in the
preparation of isocyanates and polyisocyanates; in the rubber industry as a curative for neoprene, as an anti-frosting agent (antioxidant)
in footwear; raw material in preparation of poly(amide-imide) resins (used in magnet-wire enamels); curing agent for epoxy
res ins and urethane elastomers; corrosion inhibitor; rubber additive (accelerator, antidegradant, retarder) in tires and heavy rubber
products; in adhesives and glues, laminates, paints and inks, PVC products, handbags, eyeglass frames, plastic jewelry, electric
encapsulators, surface coatings, spandex clothing, hairnets, eyelash curlers, earphones, balls, shoe soles, face masks. | | Definition | ChEBI: 4,4'-Methylenedianiline is an aromatic amine that is diphenylmethane substituted at the 4-position of each benzene ring by an amino group. It is primarily used in industry as a chemical intermediate in the production of 4,4-methylenedianiline diisocyanates and polyisocyanates.? | | General Description | A tan flake or lump solid with a faint fishlike odor. May be toxic by inhalation or ingestion, and may be irritating to skin. Insoluble in water. | | Air & Water Reactions | Oxidizes slowly in air in a reaction catalyzed by light. Somewhat hygroscopic. Insoluble in water. | | Reactivity Profile | 4,4'-Methylenedianiline polymerizes if heated above 257° F. Incompatible with strong oxidizing agents. 4,4'-Methylenedianiline is also incompatible with acids. Catalyzes isocyanate-alcohol and epoxide reactions. Flammable gaseous hydrogen may be generated in combination with strong reducing agents, such as hydrides. | | Health Hazard | TOXIC; inhalation, ingestion or skin contact with material may cause severe injury or death. Contact with molten substance may cause severe burns to skin and eyes. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution. | | Fire Hazard | Combustible material: may burn but does not ignite readily. When heated, vapors may form explosive mixtures with air: indoors, outdoors and sewers explosion hazards. Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated. Runoff may pollute waterways. Substance may be transported in a molten form. | | Flammability and Explosibility | Nonflammable | | Safety Profile | Confirmed carcinogen
with experimental tumorigenic data. Human
poison by ingestion. Poison by
subcutaneous and intraperitoneal routes.
Human systemic effects by ingestion:
rigidity, jaundice, other liver changes. An eye
irritant. Mutation data reported. It is not
rapidly absorbed through the skin.
Combustible when exposed to heat or
flame. When heated to decomposition it
emits highly toxic fumes of aniline and NOx. | | Potential Exposure | Used as an intermediate and as a curing
agent. Approximately 99% of the DDM produced is con-
sumed in its crude form (occasionally containing not more
than 50% DDM and ply-DDM) at its production site by reac-
tion with phosgene in the preparation of isocyanates and poly-
isocyanates. These isocyanates and polyisocyanates are
employed in the manufacture of rigid polyurethane foams
which find application as thermal insulation. Polyisocyanates
are also used in the preparation of the semiflexible polyure-
thane foams used for automotive safety cushioning. DDM is
also used as: an epoxy hardening agent; a raw material in the
production of polyurethane elastomers; in the rubber industry
as a curative for Neoprene and as an antifrosting agent (anti-
oxidant) in footwear; a raw material in the production of
Quana nylon; and a raw material in the preparation of poly
(amide-imide) resins (used in magnet wire enamels). | | Carcinogenicity | 4,4′-Methylenedianiline and its dihydrochloride salt are reasonably anticipated to be human carcinogens based on sufficient evidence of carcinogenicity from studies in experimental animals. | | Environmental Fate | MDA is a pale brown crystalline powder with a faint aminelike
odor. Exposure to air and light results in polymerization and
oxidation of MDA. When heated, MDA produces toxic fumes of
aniline and nitrogen oxides.
Most MDA enters the environment when it is produced or
used to make other compounds. Forty-five percent of the
produced compound is released to deep soil, 52.6% to the air,
and 2.4% to land and water. Once in the environment, it will
be mainly present as tiny particles in the air and it is removed
from the atmosphere by dry deposition, rain, and snow
scavenging. A small amount is transformed by reaction with
hydroxyl radicals. In water, most of MDA will attach to
particles and sediments, and will eventually settle to the
bottom.
The estimated half-life of biodegradation in surface water,
groundwater, and soil is 1–7 days, 2–14 days, and 1–7 days,
respectively. With respect to aquatic ecosystems, bioconcentration
factor values of 3.0–14 suggest that bioconcentration
is low, in addition this compound does not tend
to build up in the food chain. When released to soil, it is
expected to have slight to no mobility. Similarly, volatilization
from both moist and dry soil surfaces is not expected to be
important. | | Shipping | UN2651 4,40
-Diaminodiphenyl methane, Hazard
Class: 6.1; Labels: 6.1-Poisonous materials. | | Purification Methods | Crystallise the amine from water, 95% EtOH or *benzene. [Beilstein 13 IV 390.] | | Toxicity evaluation | Currently, the mechanism of action is not completely understood
and has been mostly assessed from information on
structurally similar compounds. Many of the toxic properties of
MDA have been attributed to metabolic intermediates, as these
compounds are metabolically activated by N-oxidation to
metabolites, such as N-hydroxymethylenedianiline, that react
with DNA, RNA, and proteins.
Different studies suggest that both liver and thyroid are
targets for MDA toxicity in humans and animals. Liver
toxicity has been linked to impair mitochondrial function
and structure, apoptosis, and increased oxidative stress. It
may be caused by a reactive electrophile formed during
metabolism since liver has the enzymatic routes necessary for
such activation. Experimental studies indicate that biliary
epithelial cells are damaged earlier than parenchymal cells
and bile is a major route of exposure to MDA. The mechanism
of thyroid toxicity has not yet been resolved. It has
been observed that MDA exposure can induce a slight
decrease in thyroid hormones in rats, thus triggering secretion
of thyroid-stimulating hormone (TSH), which induced
thyroid hyperplasia. Some of the induced adverse effects
observed after MDA exposure (e.g., reduced food consumption,
lower body weight gain, and effects on red cells,
lymphocytes, and clotting parameters) could be explained as
secondary responses.
MDA is carcinogenic to animals. The mechanism of liver or
thyroid tumor development remains unclear. Even if cell
injury may give indications of a nongenotoxic mechanism, it
remains still unproved, and there are also positive genotoxic
data in vitro and in vivo which indicate that a genotoxic
mechanism may be related to the formation of a reactive
metabolic intermediate cannot be excluded. Regarding thyroid
cancer, hypersecretion of TSH may have a contribution to
tumor formation. | | Incompatibilities | Dust forms and explosive mixture
with air. May polymerize in temperatures .125℃
. A weak
base. Incompatible with strong oxidizers (chlorates, nitrates,
peroxides, permanganates, perchlorates, chlorine, bromine,
fluorine, etc.); contact may cause fires or explosions. Keep
away from alkaline materials, strong acids. Flammable gas-
eous hydrogen may be generated in combination with strong
reducing agents, such as hydrides
. | | Waste Disposal | Controlled incineration
(oxides of nitrogen are removed from the effluent gas by
scrubbers and/or thermal devices). |
| | 4,4'-Methylenedianiline Preparation Products And Raw materials |
|




