|
| | N-Phenylhydroxylamine Basic information |
| Product Name: | N-Phenylhydroxylamine | | Synonyms: | N-HYDROXYANILINE;N-HYDROXYLANILINE;N-Phenylhydroxylamine;Phenylhydroxylamine;Hydroxyaminobenzene;NCI C-60093;N-Hydroxybenzenamine;N-Phenylhydroxyamine | | CAS: | 100-65-2 | | MF: | C6H7NO | | MW: | 109.13 | | EINECS: | 202-875-6 | | Product Categories: | | | Mol File: | 100-65-2.mol |  |
| | N-Phenylhydroxylamine Chemical Properties |
| Melting point | 80-84℃ | | Boiling point | 204.59°C (rough estimate) | | density | 1.1143 (rough estimate) | | refractive index | 1.5444 (estimate) | | storage temp. | -20°C | | form | solid | | pka | 9.00±0.70(Predicted) | | color | Light brown | | Water Solubility | 20g/L(5 ºC) | | Stability: | Unstable - deteriorates with storage. Incompatible with strong oxidizing agents. | | EPA Substance Registry System | Phenylhydroxylamine (100-65-2) |
| Hazard Codes | T | | Risk Statements | 25 | | Safety Statements | 45 | | RIDADR | UN 2811 | | WGK Germany | 3 | | RTECS | NC4900000 | | HazardClass | 6.1 | | PackingGroup | Ⅲ | | HS Code | 2928009090 | | Hazardous Substances Data | 100-65-2(Hazardous Substances Data) |
| | N-Phenylhydroxylamine Usage And Synthesis |
| Chemical Properties | tan powder or crystals | | Uses | Manufacture of cupferron. | | Uses | N-Phenylhydroxylamine can be used as a starting material for the synthesis of:
- 2-alkylindoles by treating with aliphatic terminal alkynes using gold catalyst via sequential 3,3-rearrangements and cyclodehydrations.
- Isoxazolidines by reacting with aldehydes and α, β-unsaturated aldehydes via a three-component one-pot catalytic reaction.
- Tetrahydro-1,2-oxazines by treating with an aldehyde and cyclopropane via homo 3+2 dipolar cycloaddition reaction.
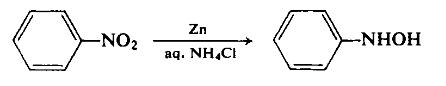
| | Definition | ChEBI: N-phenylhydroxylamine is an N-substituted amine that is a derivative of aniline in which one of the amino hydrogen atoms is replaced with a hydroxy substituent. | | Preparation | To a dispersion of 180 gm (2.75 gm-atom) of zinc dust in 500 ml of 50% aqueous ethanol, with vigorous stirring, is added 130 ml (156 gm, 1.27 mole) of nitrobenzene. The reaction is initiated by the dropwise addition of an aqueous ammonium chloride solution and can thereafter be maintained at a controllable rate by the cautious addition of the remaining ammonium chloride solution. The reaction temperature rises during this reduction step. Once reflux has subsided, the basic zinc salts are filtered off using a sintered glass funnel. The light green filtrate is cooled in an ice-salt mixture to precipitate the product. The yield is 100 gm (72.2%), m.p. 81°C.
The reduction of 2-methyl-2-nitropropane is carried out in a similar manner at 10-20°C to afford a 68% yield of N-t-butylhydroxylamine (m.p. 60-62°C).
| | General Description | Tan to brown crystals. | | Air & Water Reactions | Soluble in hot water. | | Reactivity Profile | N-Phenylhydroxylamine neutralizes acids in exothermic reactions to form salts plus water. May be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen may be generated in combination with strong reducing agents, such as hydrides. | | Fire Hazard | Flash point data for N-Phenylhydroxylamine are not available but N-Phenylhydroxylamine is probably nonflammable. | | Safety Profile | Poison by ingestion and subcutaneous routes. Human systemic effects by skin contact: primary irritation. Preparative hazard. Mutation data reported. When heated to decomposition it emits toxic fumes of NOx. | | Purification Methods | Impure base deteriorates rapidly. Crystallise it from H2O, *C6H6 or *C6H6/pet ether (40-60o). The picrate has m 186o (from EtOH), and the benzenesulfonate salt has m 70o (dec )(EtOH/*C6H6). [Beilstein 15 H 2, 15 I 3, 15, II 4, 15 III 5. 15 IV 4.] |
| | N-Phenylhydroxylamine Preparation Products And Raw materials |
|



