|
| | ILOPERIDONE Chemical Properties |
| Melting point | 118-120°C | | Boiling point | 593.7±50.0 °C(Predicted) | | density | 1.204±0.06 g/cm3(Predicted) | | Fp | 9℃ | | storage temp. | -20°C | | solubility | DMSO: soluble5mg/mL, clear | | form | powder | | pka | 8.43±0.20(Predicted) | | color | white to beige | | Merck | 14,4900 |
| | ILOPERIDONE Usage And Synthesis |
| Atypical antipsychotics | Iloperidone, an atypical antipsychotic drug, is the dual antagonist of the serotonin-2 (5-HT2) and dopamine 2 (D2). The development process of iloperidone is a bit dramatic. It is originally synthesized and identified by HE Manchester-Russell pharmaceutical companies, however, the company decided to give up further development of it, and assigned it to the Titan Pharmaceuticals; At January 1997, Titan Pharmaceuticals further transferred the goods to Novartis, and the latter one invested the III phase of the clinical development of this product and assume the registration fees of this drugs worldwide except Japan (Titan Pharmaceuticals retains the right to develop this product in Japan); however, in 2004, the Vanda Pharmaceuticals in 2004 obtain the right to develop this product from Novartis. On May 6, 2009, the US FDA approved that Vanda Pharma Company had successfully developed iloperidone (trade name: Fanapt) and make it enter into market which is mainly used for clinical treatment of adults schizophrenia.
As an orally administrated atypical antipsychotics drug, iloperidone has a obvious efficacy on reducing the positive symptoms (such as hallucinations, delusions, thought disorder, hostility, suspicion) of patients with schizophrenia as well as negative symptoms (such as unresponsiveness, emotional apathy, social apathy, of few words). Iloperidone belongs to the antagonist of 5-HT2/D2 receptor, and have a high affinity to dopamine D3 receptors. It also has appropriate affinity to the adrenergic A1 receptor, the dopamine D4 receptor, and 5-HT6 and 5-HT7 but also has a low affinity to 5-HT1A, dopamine D1 and H1 histamine receptor. Compared with currently used antipsychotics, short-term and long-term safety trials has showed that iloperidone has less adverse reactions without only minor increase of the quality of patient body. Iloperidone will not induce diabetic for the patients and also cause only a few of extrapyramidal system symptoms (such as no akathisia, no hyperprolactinemia, lower incidence of drowsiness, low decrease in cognitive decline). The clinical study has showed that iloperidone has a good safety and tolerability with good metabolism. It is mainly excreted out by the liver. The most common adverse reactions iloperidone include dizziness, dry mouth, fatigue, nasal congestion, orthostatic hypotension, lethargy, elevated heart rate, increased body mass, and prolonged QT phase. Iloperidone can also increase the risk of death in patients of Alzheimer's psychosis; thus patients of Alzheimer's psychosis are not allowed to use this drug.
The above information is edited by the Chemicalbook of Dai Xiongfeng.
| | Physical and chemical property | This product is white or off-white crystalline powder which is almost insoluble in water; slightly soluble in hydrochloric acid; soluble in chloroform, ethanol, methanol and acetonitrile.
| | Pharmacological effects | Iloperidone is a derivative of piperidinyl oxacillin and has a high affinity (IC50 = 9.3nmol /L) to 5-HT2 receptors in rodents but has a one-order magnitude lower affinity to D2 receptor (IC50 = 109nmol/L); It also has a very high affinity to the adrenergic a1 receptors (IC50 = 0.4 nmol/L), but has much lower affinity to a2, 5-HT1A, O and D1 receptors affinity (IC50: 60, 210, 180 and 750nmol/L, respectively). This product doesn’t bind to the muscarinic acetylcholine cholinergic receptors and N-methyl-D-aspartate ion channel site.
In vivo or in vitro experiments have showed that the drug has similar pharmacological properties as other atypical antipsychotics, namely selective take effects on mesolimbic areas and anti-psychotic symptoms, and thereby causing a low incidence of EPS.
In vitro autoradiography study of receptor found that continuous intraperitoneal injection 5mg/kg dose of the product for 19 days to rats can significantly reduce the number of 5-HT2 receptors in frontal cortex, but affect neither the receptor affinity nor the number and affinity of D2 receptors of six regions in nucleus accumbens and striatum. This is the significant difference between this product and haloperidol with the latter one being able to increase the number of D2 receptors in these regions (this may be one of the factors for causing its high incidence of EPS).
Study on the effects of this product on the agonist activity of lysergic diethylamide (LSD) in HEK-293 and CHO-K1 cells expressing human D2A receptors and human a2C has showed that, the agonist activity of LSD on D2A receptors and a2C adrenergic receptor (pIC50 were 8.69 +/-0.08 and 8.73 +/-0.05, respectively) can be completely blocked [pKB were (8.684 +/-0.14) and (8.13 +/-0.03), respectively] by this product. Therefore, blockage of a2C adrenoceptor may enhance its antipsychotic effects induced by blocking D2 receptor.
Radioligand binding assays show that this good has a high affinity on a1-adrenergic receptors, and D3 and 5-HT2A receptor, indicating that it is the only potential broad-spectrum antipsychotics with few side effects.
| | Synthetic method | Use difluorobenzene as the starting material, go through Friedel-Crafts reaction, then react with hydroxylamine into oxime and go through the intramolecular substitution reaction in the presence of a strong base sodium hydride to form a ring, then react with 1-[4-(3-chloropropyl)-3-methoxyphenyl] ethanone to obtain iloperidone.

Figure 1 the artificial synthesis route of iloperidone
| | Drug Interactions | 1. This product is not the substrate of CYP1A1, CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19 or CYP2E1, so this product won’t have interaction when combined with either inducers or inhibitors of these enzymes. This product is mainly metabolized by CYP3A4 and CYP2D6. The inhibitors of either CYP3A4 (e.g., ketoconazole) or CYP2D6 (such as fluoxetine, paroxetine) can inhibit the metabolism of the product and increase its plasma concentration.
2. In vivo test of the human liver microsomes have showed that this product has on inhibitory effects on the drugs which are metabolized by the CYP1A1, CYP1A2, CYP2A 6, CYP2B6, CYP2C8, CYP2C9, or CYP2E1 enzyme; It also has no induction effect on CYP1A2, CYP2C8, CYP2C9, CYP2C19, CYP3A4 and CYP3A5 enzymes.
3. This product should be avoided being combined with QT-prolonging drugs including class IA antiarrhythmics drugs (quinidine, procainamide) or Class III antiarrhythmic drugs (amiodarone, sotalol); antipsychotics drugs (chlorpromazine, thioridazine); antibacterial drugs (gatifloxacin, moxifloxacin); other drugs (pentamidine, acetyl L-methadone). This product should be avoided from being used in congenital prolonged-QT phase patients and patients with arrhythmia history.
| | Adverse reaction | Compared with currently used antipsychotics, short-term and long-term tests have showed that this product has few adverse reactions, increase the quality of patient body to a small extent,. Iloperidone doesn’t nduce diabetic for the patients and also cause only a few of extrapyramidal system symptoms (such as no akathisia, no hyperprolactinemia, lower incidence of drowsiness, low decrease in cognitive decline). The clinical study has showed that iloperidone has a good safety and tolerability with good metabolism. It is mainly excreted out by the liver. The most common adverse reactions iloperidone include dizziness, dry mouth, fatigue, nasal congestion, orthostatic hypotension, lethargy, elevated heart rate, increased body mass, and prolonged QT phase. Iloperidone can also increase the risk of death in patients of Alzheimer's psychosis; thus patients of Alzheimer's psychosis are not allowed to use this drug.
| | Intellectual Property and Patents | There are two patents related to domestic iloperidone with both applicants being the Novartis AG companies. One is a depot agent of iloperidone and star polymers which is applied on 2002 but without being authorized. It belongs to the composition patent of a special formulation and is relevant to reporting iloperidone and its tablets; the other is the patent about new application of iloperidone applied in 2002 and authorized at 2005. The protection scope is related to the application of the iloperidone in treating affective disorder including bipolar affective disorder, but is not related to schizophrenia. It also doesn’t affect the declaration of iloperidone and its tablets.
| | Description | Iloperidone is an atypical antipsychotic and adrenergic, dopamine, and serotonin (5-HT) receptor antagonist. It binds to several receptors, including the α1-adrenergic receptor (α1-AR), α2-AR, and dopamine D2 receptor (Kis = 0.31, 3, and 3.3 nM, respectively), as well as the 5-HT1A, 5-HT1D, 5-HT2A, and 5-HT2C receptors (Kis = 33, 15, 0.2, and 14 nM, respectively) in radioligand binding assays using human post-mortem brain tissue. Iloperidone also binds to human D1, D3, D4, D5, and rat 5-HT2 receptors (Kis = 216, 7.1, 25, 319, and 3.1 nM, respectively, in CHO cells) and the histamine H1 receptor (Ki = 12.3 nM in human post-mortem brain tissue). Iloperidone (1-3 mg/kg) prevents the reduction in prepulse inhibition induced by apomorphine , phencyclidine (PCP), and cirazoline in rats. It also increases the time rats spend in the open arms of the elevated plus maze and the number of social interactions when administered at a dose of 0.5 mg/kg. Formulations containing iloperidone have been used in the treatment of schizophrenia. | | Chemical Properties | Off-White Solid | | Uses | Combined dopamine (D2) and serotonin (5HT2) receptor antagonist. Antipsychotic. | | Uses | Iloperidone (Fanapt, Fanapta, Zomaril) is an atypical antipsychotic for the treatment of schizophrenia. Iloperidone is a monoamine directed towards acting upon and antagonizing specific neurotransmitters, particularly multiple dopamine and serotonin recep | | Definition | ChEBI: A member of the class of piperidines that is the 4-acetyl-2-methoxyphenyl ether of 3-(piperidin-1-yl)propan-1-ol which is substituted at position 4 of the piperidine ring by a 6-fluoro-1,2-benzoxazol-3-yl group. A member of the group of second generation a
tipsychotics (also known as an atypical antipsychotics), it is used for the treatment of schizophrenia. | | General Description | Iloperidone is an atypical antipsychotic administered for the treatment of schizophrenia. The drug is marketed under the trade names Fanapt and Fanapta. This Certified Spiking Solution? is suitable for use in LC/MS or GC/MS applications including clinical toxicology, forensic testing, or pharmaceutical research. | | Biochem/physiol Actions | Iloperidone is an atypical antipsychotic, a combined dopamine (D2) and serotonin (5HT2) receptor antagonist. Iloperidone targets a selective set of dopamine, serotonin and also norepinephrine receptor subtypes. Like similar atypical antipsychotics, Iloperidone has higher affinity for the 5-HT2A receptor (Ki < 10 nM) than for the D2 receptor (Ki = 10–100) nM). Iloperidone has high affinity for dopamine D3 (Ki < 10 nM) and lower affinity for other serotonin or dopamine subtypes. Unlike most atypical antipsychotics, Iloperidone also has high affinity for norepinephrine alpha1 receptors (Ki < 10 nM) which may contribute to the drug′s efficacy on mood and cognition. | | Clinical Use | Acting as an antagonist on serotonin (5-HT2) and dopamine receptor subtypes, iloperidone is an
antipsychotic indicated for the treatment of acute schizophrenia in adults. Based on its in vitro and in
vivo binding properties against both serotonin and dopamine receptors, it is expected that iloperidone
will show fewer extrapyramidal symptoms than currently marketed antipsychotics such as haloperidol
and clozapine. The original discovery was made by Hoechst-Roussel Pharmaceuticals who passed
the developing rights to Vanda Pharmaceuticals and subsequently Novartis for marketing in the U.S.
and Canada. While this drug was originally approved in the U.S. in 2009, the marketing was only
initiated in the U.S. in 2010. | | Synthesis | Although a number of syntheses are reported in the literature, the process
enabled route from a key intermediate 98 is described in the scheme. The key intermediate 98
was synthesized from isonipecotic acid (94) in four steps. Formylation of isonipecotic acid (76%
yield) followed by conversion of the acid to the acyl chloride gave 95 in 100% yield. Friedel-Crafts
acylation of 1,3-difluorobenzene (96) with the acid chloride 95 provided ketone 97 in 32% yield. Treatment of ketone 97 with hydroxylamine hydrochloride in the presence of potassium hydroxide gave the corresponding oxime, which upon refluxing in 2-ethoxylethanol and water cyclized to
piperidine benzisoxazole 98 with concomitant loss of the N-formyl group. Alkylation of piperidine 98
with 1-chloro-3-bromo propane in DMF in the presence of potassium carbonate provided the chloride
intermediate 99 in 80% yield. Subsequent reaction with phenol 100 under basic conditions gave the
desired product iloperidone (VII) in 81% yield. 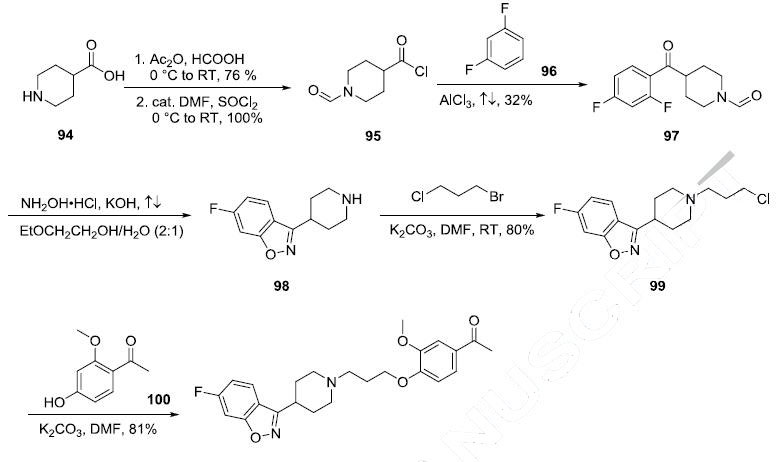
|
| | ILOPERIDONE Preparation Products And Raw materials |
|